
Announcement: OGI Genetic Eye Disease (GEDi) Diagnostic Test Panels and Pricing Have Been Updated As of 11/1/2016
GENETIC EYE DISEASE (GEDi) DIAGNOSTIC TEST INFORMATION:
TESTING PANELS:
- Genetic Eye Disease Panel for Retinal Genes (GEDi-R):
Two-hundred, sixty-seven genes (see gene list below) currently known to be involved in inherited retinal degenerations and related disorders are analyzed by a combination of SureSelect targeted enrichment, followed by Next Generation sequencing using an Illumina MiSeq instrument. This test also includes sequencing of the mitochondrial genome. The following intronic variants are analyzed: CEP290 c.2991+1655A>G; PRPF31 c.1374+654C>G; USH2A c.7595-2144A>G; OFD1 c.935+706A>G, 5 ABCA4 (c.5196+1137G>A, c.5196+1216C>A, c.5196+1056A>G, c.4539+2001G>A, c.4539+2028C>T), CHM c.315-4587T>A, SDCCAG8 c.740+356C>T, PRDM13 chr6:100040906G>T; chr6:100040987G>C; chr6:100041040C>T.
- Genetic Eye Disease Panel for Optic Nerve Disease and Early Onset Glaucoma (GEDi-O):
Twenty-two genes (ACO2, AFG3L2, AUH, C12orf65, CISD2, CYP1B1, FOXC1, FOXF2, LTBP2, MTPAP, MYOC, NDUFS1, NR2F1, OPA1, OPA3, OPTN, PAX6, PITX2, POLG, SPG7, TEK, and WFS1) currently known to be involved in optic nerve disease, early onset glaucoma and related disorders are analyzed by a combination of SureSelect targeted enrichment, followed by Next Generation sequencing using an Illumina MiSeq instrument. Panel also includes sequencing of the mitochondrial genome.
- Genetic Eye Disease Panel for Strabismus (Gedi-S):
Eight genes (ROBO3, PHOX2A, HOXA1, SALL4, CHN1, TUBB3, KIF21A, HOXB1) currently known to involved in congenital cranial dysinnervation disorders and disorders of axon guidance analyzed by a combination of SureSelect targeted enrichment followed by Next Generation sequencing using an Illumina MiSeq instrument.
- Confirmation of Research Findings or Familial Variant Testing:
Sanger sequencing is performed to confirm a genetic variant identified by research testing or confirmation of a familial variant. For this testing, please provide the gene name and variant information (genomic position, cDNA, and protein information).
Please note: Turn-around time is 90 Days.
GEDi GENE LIST:
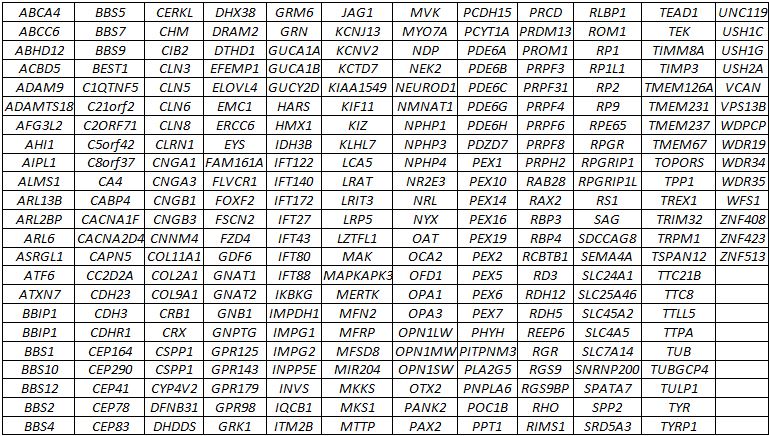
METHODOLOGY AND TECHNICAL INFORMATION:
For each panel, the coding regions and splice regions of genes tested are examined. Sanger sequencing is performed to confirm all clinically significant variants and, when necessary, to fill in regions of insufficient coverage.
This testing will not detect large genomic structural rearrangements such as deletions, duplications, inversions, and insertions. Variants in non-coding regions, which are outside of the splicing regions and not specifically targeted, will not be analyzed. Additionally, genetic variants present in genes not known to be associated with retinal degeneration may not be detected by this testing method. Research-based testing is available for detecting these types of genetic alterations.
ORDERING TESTING:
Genetic testing can only be ordered by a medical professional.
Please complete the updated Diagnostic Test Requisition Form and mail the original copy to the shipment address listed below.
This completed form MUST accompany all specimens (however, if the sample is drawn at a separate facility, the form can be sent to the address below separately, but the sample should be sent in with a copy of the first page of the form).
The Test Requisition includes the following:
- Patient, testing, and sample information (Pages 1-2)
- Consent form (Page 3)
- Billing information (Page 4)
A specimen cannot be processed without a completed requisition form.
SPECIMEN AND SHIPMENT REQUIREMENTS:
Blood Specimens are the preferred specimen type for testing.
- Volume for children: 5-8 mL of whole sterile blood collected in EDTA (lavender top) tube.
- Volume for adults: 8-10 mL of whole sterile blood collected in EDTA (lavender top) tube.
- Labeling: Tubes should be labeled with the patient’s name, date of birth, and/or ID number.
- Shipping: Specimens should be shipped at room temperature and by overnight delivery with an arrival of Monday-Friday. Samples should be received within 48 hours of collection. NO WEEKEND DELIVERY. Samples can be refrigerated before shipping. DO NOT FREEZE BLOOD. In hot weather, a cool pack can be used. Shipping costs are the responsibility of the sender.
SAMPLES SHOULD BE SHIPPED TO THE FOLLOWING ADDRESS:
Ocular Genomics Diagnostic Laboratory
Massachusetts Eye and Ear Infirmary
243 Charles Street, Room 566A
Boston, MA 02114
PRICING:
| Test | Price | CPT Code |
| GEDi-R | $2850 | 81434* |
| GEDi-O | $1650 | 81479 |
| GEDi-S | $1250 | 81479 |
| Validation/Segregation Testing | $500 | 81403 |
CLIA Certification Number: 22D1037777 *Please note that the CPT Code for GEDi-R Test has changed to 81434 (formerly 81479).
BILLING AND PAYMENT INFORMATION:
Payment Policy:
As a not-for-profit diagnostic laboratory, we cannot accept commercial or managed care insurance, including Medicare and out-of-state Medicaid, unfortunately. Please note that we also do not exchange any communication with insurance organizations. We will do all we can to ensure that you have the documentation necessary to complete an insurance reimbursement claim and will provide you with an itemized receipt when the test results are available and payment received.
Please note: If we receive a sample in the lab without payment, or incomplete billing information, we will hold the sample and begin testing once receive payment or the required institutional billing information.
Institutional Billing:
OGI offers a number of billing options for outside institutional clients and their patients; however, we do not bill insurance companies and are unable to begin testing without accurate billing information. We can provide a detailed receipt and document of services provided to clients with patients seeking reimbursement from their insurance company as mentioned above, but please note that this is the extent to our involvement in the process. Please note: Our policy is to bill after beginning testing and payment should be remitted within 45 days of receipt of bill. Test results will be sent after the approximate 90-day testing turn-around time frame.
*Please include the facility name, address, billing contact person, email, fax, and phone number on the requisition form. OGI accepts checks (payable to Massachusetts Eye and Ear Infirmary) and credit card payments.
Patient Self-Pay:
We offer several billing options to our clients and their patients; however, as mentioned above, we do not bill insurance companies for patients and are unable to begin testing without accurate billing information.
For direct patient billing, payment must be received before testing can begin. We accept checks (payable to Massachusetts Eye and Ear Infirmary) and credit cards. The client is responsible for collecting accurate and up-to-date credit card information from their patients. We do not interface with our clients’ patients. There is a $20.00 processing fee for insufficient check payments.
For Patients Seeking Reimbursement from an Insurance Company:
It is the responsibility of the patient to seek reimbursement from their insurance company. Every insurance company has different policies and required documents. We advise you to make contact with your insurer before submitting a sample for testing.
You need:
- The name, address and telephone number of the clinician recommending testing
- The name of the genetic test being recommended
- CPT Codes (see Diagnostic Test pricing chart)
You should ask:
- Will I need pre-authorization?
- Will I need a Letter of Medical Necessity?
- What is the reimbursement process?
- What will I need to submit for reimbursement?
- What forms are required? Where can I find those forms?
- Where do I submit any paperwork?
For inquiries, please contact: OGI_Diagnostics@meei.harvard.edu.
Pricing updated 11/4/2016
